

 |
 |
ElectrochemistryStep 1: -Investigating redox reactions -Practice with redox reactions -Reduction tendencies of metal ions Step 2: Step 3: Step 4: -Practice with non-standard cells Feedback Form: |
Electrochemistry Tutorial: Galvanic Cells and the Nernst Equation >> Introduction
Electrochemistry: Galvanic Cells and the Nernst EquationIntroduction
In this experiment:
The experimental strategy is shown below in the following 4 steps:
Step 1 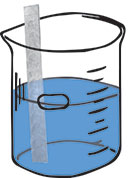 Action: Dip a strip of metal in a salt solution of another metal. Repeat with different metal/salt combinations. Result: The observations provide evidence of whether or not a spontaneous reaction takes place. If a reaction is observed, we can use our knowledge of the chemical species involved to figure out the chemical reaction.
Step 2
Result: We identify the galvanic cells which provide enough voltage to power a stopwatch.
Step 3
Result: Create a series of electrode potentials arranged in order of the tendency of a metal ion to undergo reduction.
Step 4
Result: We determine the values of cell voltage under non-standard conditions. The following pages contain tutors to assist you with understanding and applying some concepts required in the above steps. Please click next to continue to the online activity.
|
| Page Last Updated: 06.22.2021 |