Stoichiometry Bridge Course
The ChemCollective, in collaboration with Carnegie Mellon's Open Learning Initiative (OLI), is offering free access to an online course in stoichiometry. The course is aimed at:

Students measure arsenic in well water
- High school students who would like additional help and practice with stoichiometry concepts and problem solving
- Students about to enter AP or college chemistry who would like a review of stoichiometry
Topics covered
The course begins with foundation topics such as dimensional analysis, significant figures, the mole, and molecular weight (molar mass.) This is followed by solution, composition and reaction stoichiometry. The most advanced topics are empirical formula, limiting reagents, titrations, and using reactions to determine the composition of mixtures. You can easily customize the syllabus to offer your students any subset of these topics.
Before taking the course, students need only have a basic familiarity with the meaning of chemical formulas, such as H2O, and reactions, such as that for the burning of Hydrogen in air: 2H2 + O2 --> 2H2O.
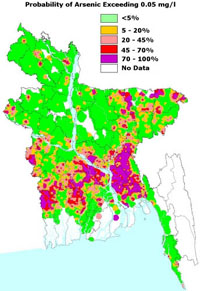
Arsenic levels in Bangladesh
Set in a real-world scenario
The course employs the backdrop of Arsenic contamination in the ground water of Bangladesh, which allows students to learn and apply their knowledge in the context of real world issues. Activities include determining if a water sample is safe to drink, determining the form of arsenic is a ground sample, and characterizing the ability of various powders to absorb arsenic. As the course progresses, the emphasis shifts towards the challenges facing modern analytical chemistry, especially the difficulty of affordably detecting small amounts of arsenic in well water. The scenario gives students a memorable application of the core topics of stoichiometry.
Course features
Each topic of the course includes an introductory movie and some interactive problems with hints. These serve as interactive worked examples since reading all the hints walks you through how to do the problem. Most topics conclude with either a virtual lab, that allows students to apply their knowledge by designing and carrying out an experiment or a randomized activity that gives help and feedback as students solve a challenging problem.
Getting started
Open access version: [http://oli.web.cmu.edu/jcourse/webui/guest/look.do?section=chemistry] You can try the course now without even creating a login. This will allow you to see the entire course, but will not record your progress in the gradebook. If you would like the system to remember who you are when you return, please sign up for a free account and, when asked for your course admit code, enter 'stoichdemo'.
Use this course in your classroom. If you would like to utilize the OLI gradebook and assessment options to keep track of your students' progress, please go to: https://oli.web.cmu.edu/jcourse/webui/signup/instructor.do to register for an instructor account. Shortly after you have registered, you will receive email that will walk you through the process for setting up a class section and using the features of the OLI system. If you have any questions, please feel free to email us at stoich@chemcollective.org.
Please help us improve the course
In collaboration with the Pittsburgh Science of Learning of Center (http://www.learnlab.org), we are actively seeking classrooms that will work with us to determine the effectiveness of the course. Participation can range from simply providing feedback on how the course was or was not useful in your classroom to allowing us to anonymously monitor student learning in course. Professional development opportunities in the Science of Learning Center include attending workshops on how to effectively use, and potentially develop, online course materials.